By SPC Staff
The FDA approved levoketoconazole (Recorlev, Xeris Biopharma Holdings) for the treatment of endogenous hypercortisolemia in adult patients with Cushing’s syndrome for whom surgery is not an option or has not been curative.
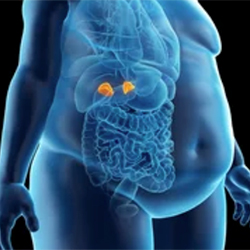
The approval of levoketoconazole, a cortisol synthesis inhibitor, was based on safety and efficacy data from two phase 3 studies that evaluated a combined study population of 166 patients, which was representative of the adult drug-treated U.S.
