By SPC News Staff
The FDA approved the combination of durvalumab (Imfinzi, AstraZeneca) and tremelimumab (Imjudo, AstraZeneca) plus platinum-based chemotherapy for the treatment of adult patients with stage IV non-small cell lung cancer (NSCLC).
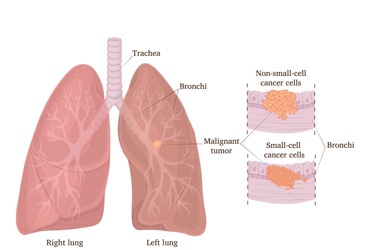
The approval was based on the results from the POSEIDON phase 3 trial. Patients treated with a limited course of five cycles of the anti-CTLA-4 (cytotoxic T lymphocyte–associated antigen-4) antibody durvalumab plus tremelimumab and four cycles of
