new drug approvals
ASX23: Specialty Drugs Dominating New FDA Approvals in 2023
Specialty drugs continue to be the main category in the FDA’s pipeline for new approvals heading into ...
MAY 3, 2023

FDA Approves Oral Form of Radicava to Treat Adults With ALS
ALS is a rare disease that attacks and kills the nerve cells that control voluntary muscles. The CDC estimates ...
MAY 13, 2022
FDA Grants New Indication to Taltz for Certain Axial Spondyloarthritis
Ixekizumab is a monoclonal antibody that inhibits the release of proinflammatory cytokines and chemokines by ...
JUNE 3, 2020
2017 in Review: Fighting for the Specialty Market & Pt Journey
Every year a 2012 statement made by Phil Hagerman, RPh, the CEO at Diplomat, becomes a little truer: Specialty is ...
DECEMBER 30, 2017

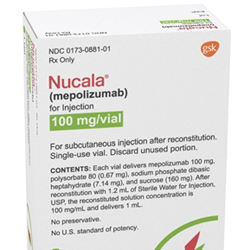
FDA Approves New Indication for Nucala
The FDA expanded the approved use of Nucala to treat adults with eosinophilic granulomatosis with polyangiitis, a ...
DECEMBER 14, 2017
Forewarned Is Forearmed: Monitoring the Drug Pipeline
SPC Editorial Director David Bronstein talks with Nicole Trask, PharmD, a consultant pharmacist at Commonwealth ...
OCTOBER 18, 2017

FDA Approves Besponsa for Adults With Relapsed or Refractory ALL
Pfizer's new drug is the first and only CD22-directed antibody–drug conjugate indicated for the treatment of ...
AUGUST 17, 2017
GAO: FDA Lacks Data To Track Expedited Drugs Post Approval
The GAO questions the FDA's ability to oversee hundreds of approved drugs with fast-track designations.
JANUARY 25, 2016

